CAR T cell therapy uses a lab-engineered protein grafted onto a patient’s own T cells that directs them to seek and destroy cancer cells bearing a specific target on their surface. It has transformed leukemia treatment, but more than half of patients eventually relapse—often because leukemia cells shed the surface antigens that CAR T cells are engineered to recognize.
Researchers at the Institute of Process Engineering (IPE), Chinese Academy of Sciences, working with Zhujiang Hospital and the Institute of Hematology & Blood Diseases Hospital, have now developed a biomimetic platform that keeps CAR T cells effective even when antigen levels collapse, without any additional genetic modification of the cells.
Published in Cell (doi: 10.1016/j.cell.2026.02.005) on March 9, 2026, the approach centers on a protein called ferritin—the natural binding partner of CD71, an iron-transport receptor found abundantly on both CAR T cells and leukemia cells across disease types and stages. By precisely controlling assembly conditions, the team induced ferritin to self-assemble into a molecular bridge they call FACE (ferritin aggregation cell engager). Added to CAR T cells during routine preparation, FACE latches onto CD71 on the T cell surface; after infusion, it simultaneously grabs CD71 on leukemia cells, physically locking the two together and sharpening the immune attack. In patient-derived mouse models with normal antigen levels, FACE-CAR T cells matched the efficacy of conventional CAR T cells at just one-fifth the dose, reducing the risk of cytokine release syndrome. When antigen expression fell below 10% of normal—a level at which standard CAR T cells largely fail—FACE-CAR T cells still cleared the cancer and achieved 100% survival of model mice. Loading ferritin’s hollow cage structure with chemotherapy drugs produced a further-enhanced version, FACED, which eliminated even antigen-negative leukemia cells—the subpopulation most responsible for relapse. Because FACE is made from an endogenous protein and FDA-approved polymer derivatives, it can be incorporated into existing CAR T manufacturing as a simple culture supplement, with no new engineering required.
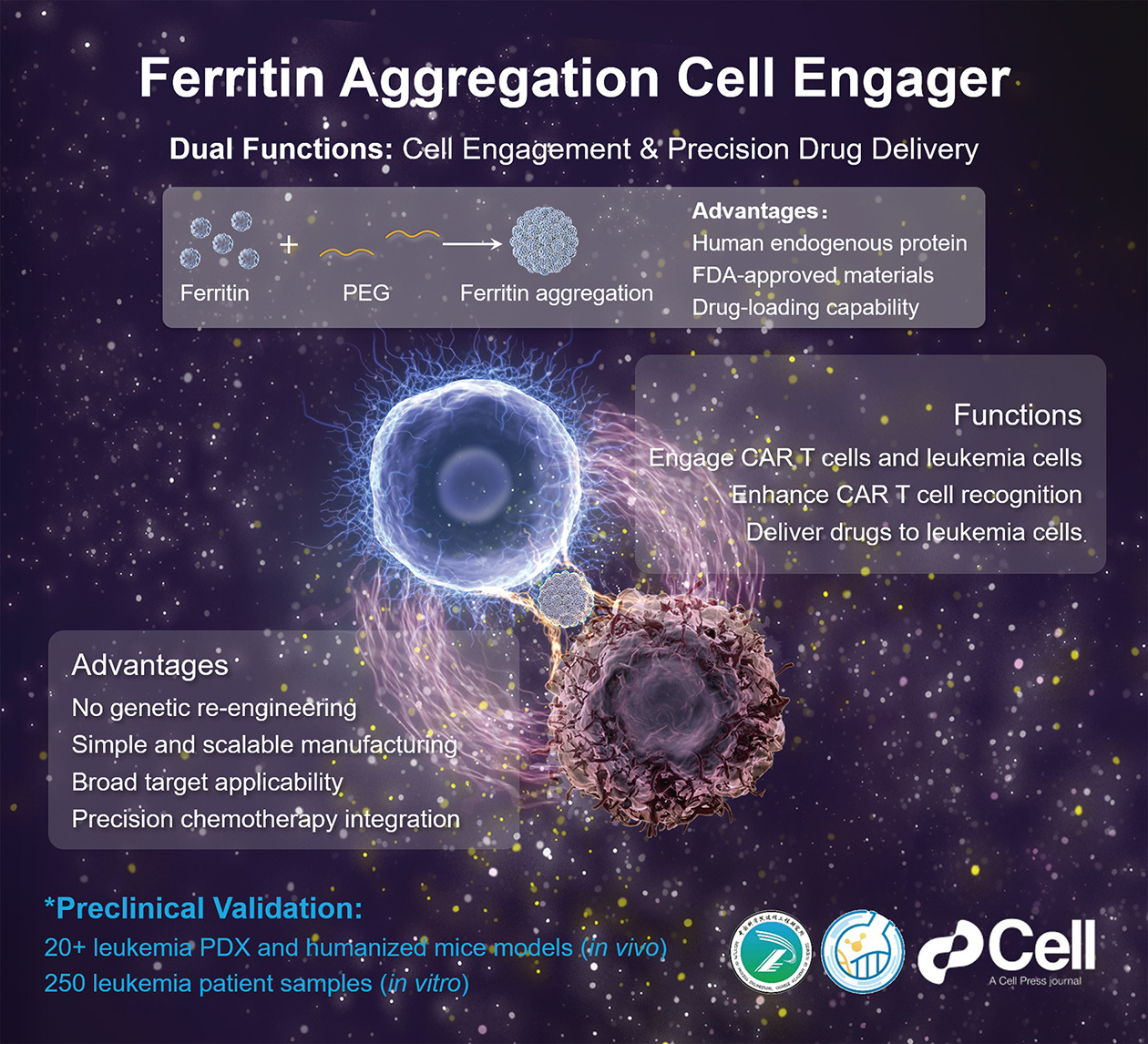
A biomimetic platform using ferritin aggregation as a bridge to enhance CAR T cell therapy against leukemia. (Image by LI Feng)

